8th Grade Science
Year in Review
| Year in Review.docx |
Magnets + Electricity
Read:
- pg. 404-405
- pg. 411-412
Answer:
- pg. 409 # 2, 4, 5
- pg. 421 # 1
- pg. 426 # 9, 10, 11, 12
Read:
- pg. 404-405
- pg. 411-412
Answer:
- pg. 409 # 2, 4, 5
- pg. 421 # 1
- pg. 426 # 9, 10, 11, 12
Law of Conservation of Mass
| Law of Conservation of Mass.pdf |
Practice TCAP
| Practice TCAP 8th Science.pdf |
Input Answers to Practice Test
Online Textbook
Click HERE to access the online textbook.
The link is http://glencoe.mheducation.com/sites/0078901367/ or also bit.ly/2G2vXS9
The link is http://glencoe.mheducation.com/sites/0078901367/ or also bit.ly/2G2vXS9
Acids & Bases
In the textbook, read pgs. 322 - 328 (skip 327).
1. Why are some acids safe to eat but others are not?
2. List 4 household/common acids.
3. What kind of ions do bases attract?
4. List 4 household/common bases.
5. Estimate the pH of lemon juice. Justify your answer.
6. Substance X has a pH of 8. Substance Y has a pH of 11. Substance Y is ___ times more basic than Substance X.
7. Describe what happens to litmus paper when added to an acid, to a base, and to water.
1. Why are some acids safe to eat but others are not?
2. List 4 household/common acids.
3. What kind of ions do bases attract?
4. List 4 household/common bases.
5. Estimate the pH of lemon juice. Justify your answer.
6. Substance X has a pH of 8. Substance Y has a pH of 11. Substance Y is ___ times more basic than Substance X.
7. Describe what happens to litmus paper when added to an acid, to a base, and to water.
States of Matter & Changes of State
| States of Matter.pdf |
Read through pg. 15 - 27 and write down answers as you go.
1. What is easier to boil: alcohol or water? How do you know?
2. Justify sugar being classified as a solid. Justify sugar being classified as a liquid.
3. Give 5 examples of aerosols.
4. Explain why a breeze helps us cool down when we're outside in hot weather.
5. What would happen if a jet engine were made of steel?
6. If you placed carbon dioxide, methane, oxygen, and nitrogen into an environment that was -250* F, which would be gases? Which would be liquids? Which would be solids?
Read through pg. 34 - 37 ("Particle Theory")
1. Using the arrangement of atoms, explain why you can put your hand through a cloud of steam and a bowl of liquid water, but not through a block of ice.
2. How does a solid reach its melting point? Describe what happens to the atoms and molecules.
3. Go back to pg. 27 and read the paragraph about "Dry Ice"...draw a diagram (similar to the ones on pg. 34/35) of what happens to the atoms when solid carbon dioxide sublimes.
4. What kind of temperature change occurs when gaseous water (water vapor) turns to liquid water (water droplets)?
5. Combine the ideas of Particle Theory and the property of density to explain why solids weigh more than liquids, which weigh more than gases.
1. What is easier to boil: alcohol or water? How do you know?
2. Justify sugar being classified as a solid. Justify sugar being classified as a liquid.
3. Give 5 examples of aerosols.
4. Explain why a breeze helps us cool down when we're outside in hot weather.
5. What would happen if a jet engine were made of steel?
6. If you placed carbon dioxide, methane, oxygen, and nitrogen into an environment that was -250* F, which would be gases? Which would be liquids? Which would be solids?
Read through pg. 34 - 37 ("Particle Theory")
1. Using the arrangement of atoms, explain why you can put your hand through a cloud of steam and a bowl of liquid water, but not through a block of ice.
2. How does a solid reach its melting point? Describe what happens to the atoms and molecules.
3. Go back to pg. 27 and read the paragraph about "Dry Ice"...draw a diagram (similar to the ones on pg. 34/35) of what happens to the atoms when solid carbon dioxide sublimes.
4. What kind of temperature change occurs when gaseous water (water vapor) turns to liquid water (water droplets)?
5. Combine the ideas of Particle Theory and the property of density to explain why solids weigh more than liquids, which weigh more than gases.
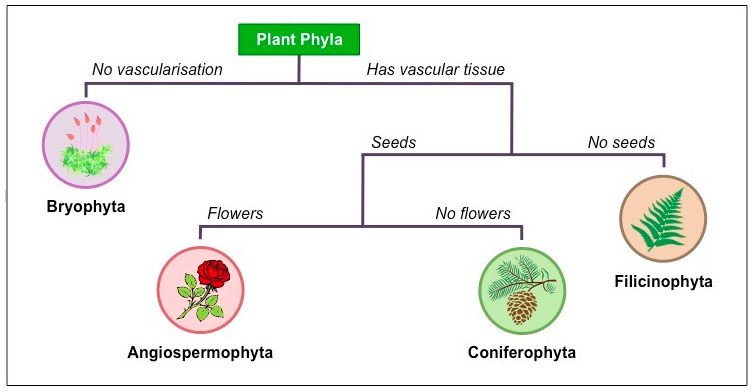
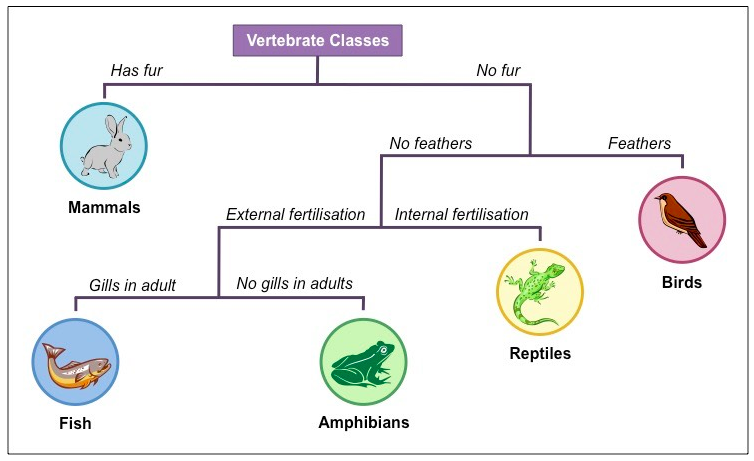
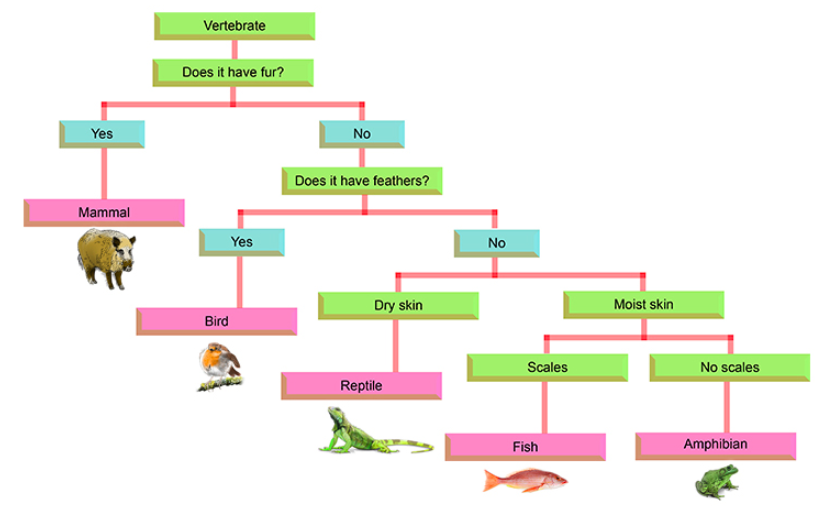
Dichotomous Keys
| Dichotomous Key 1.pdf |